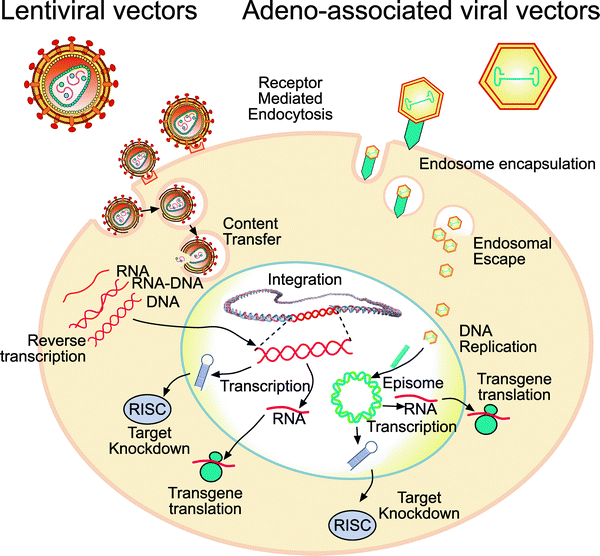
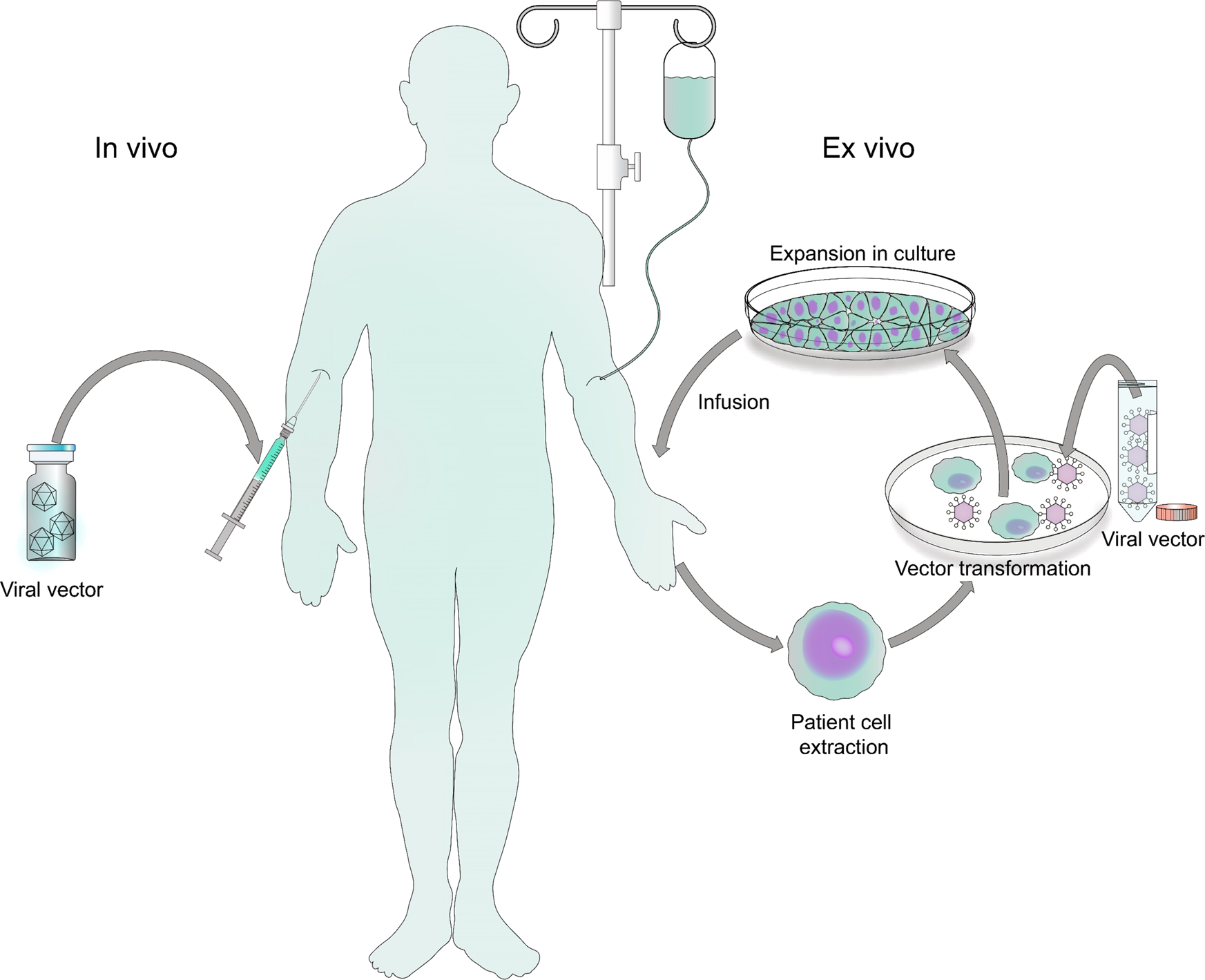
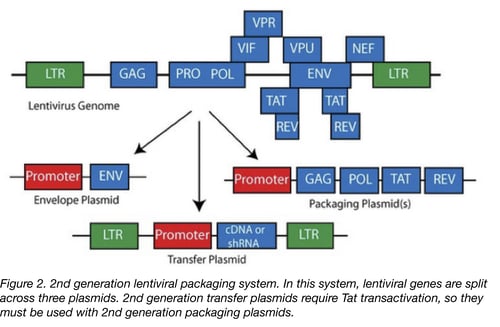
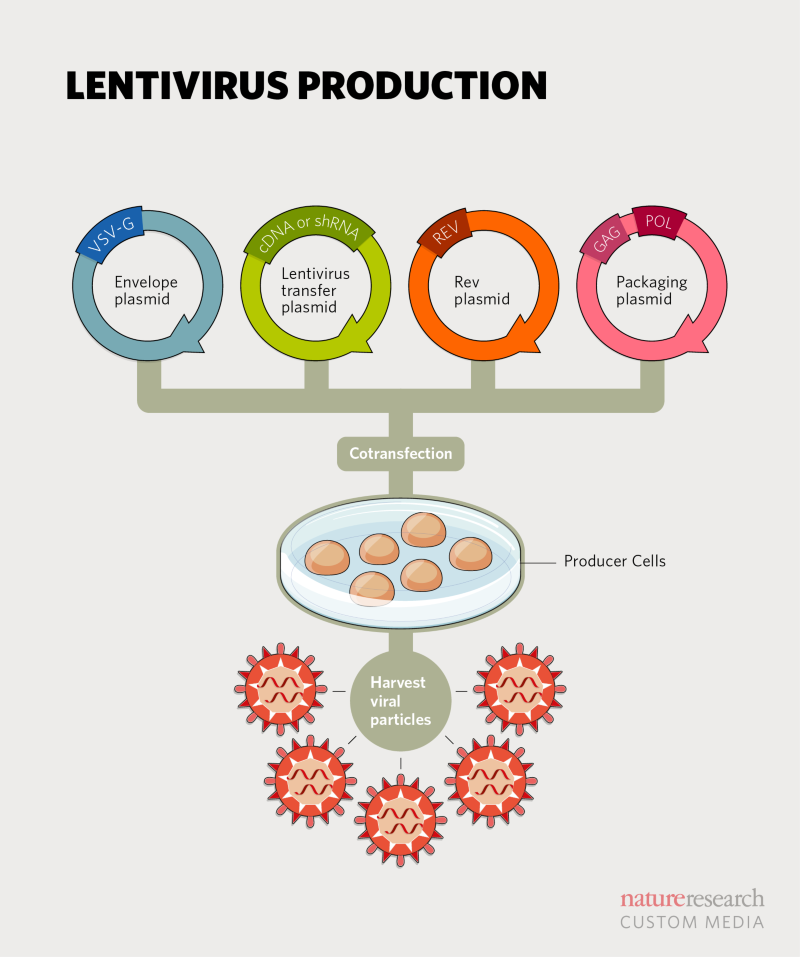
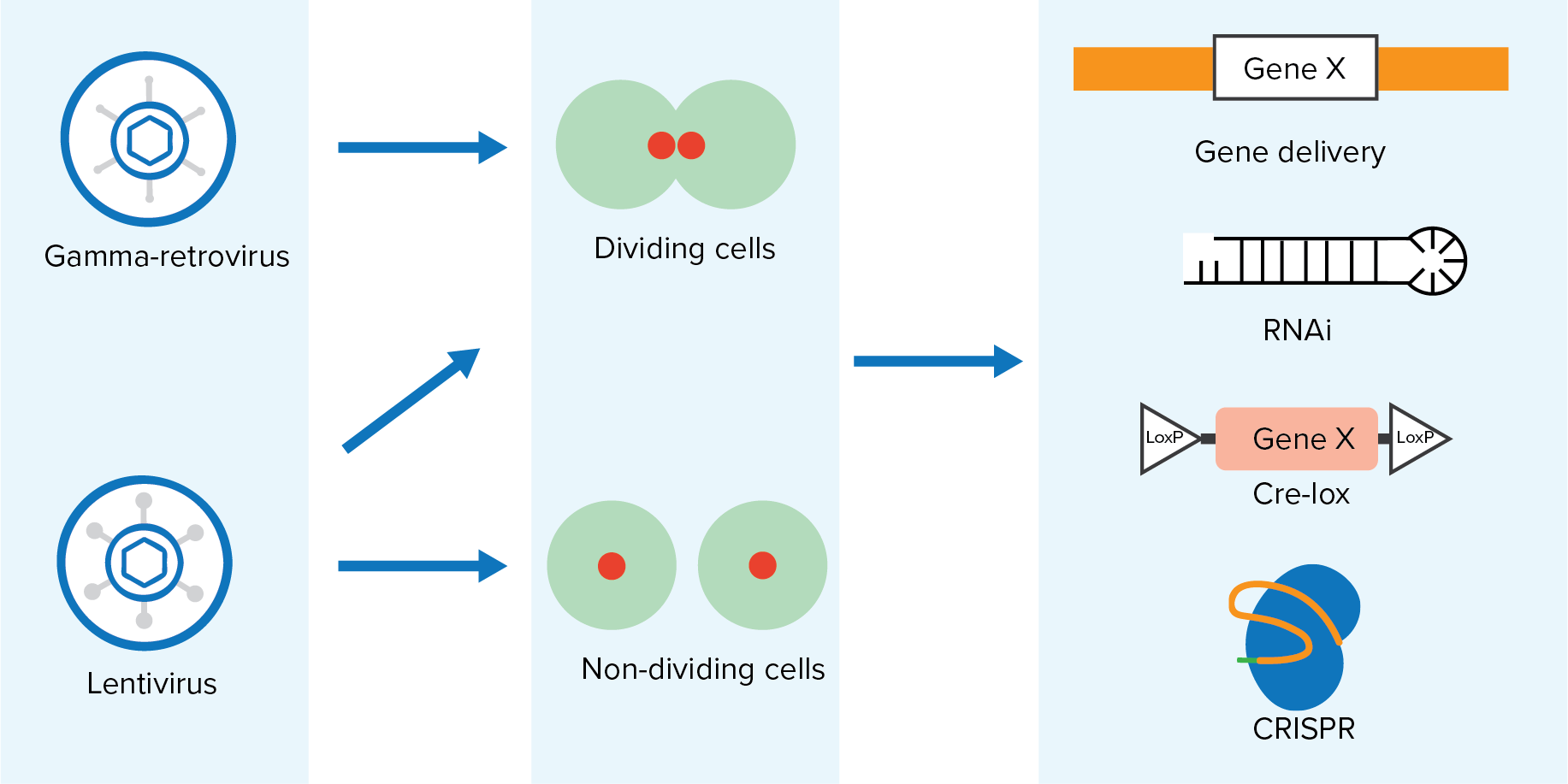
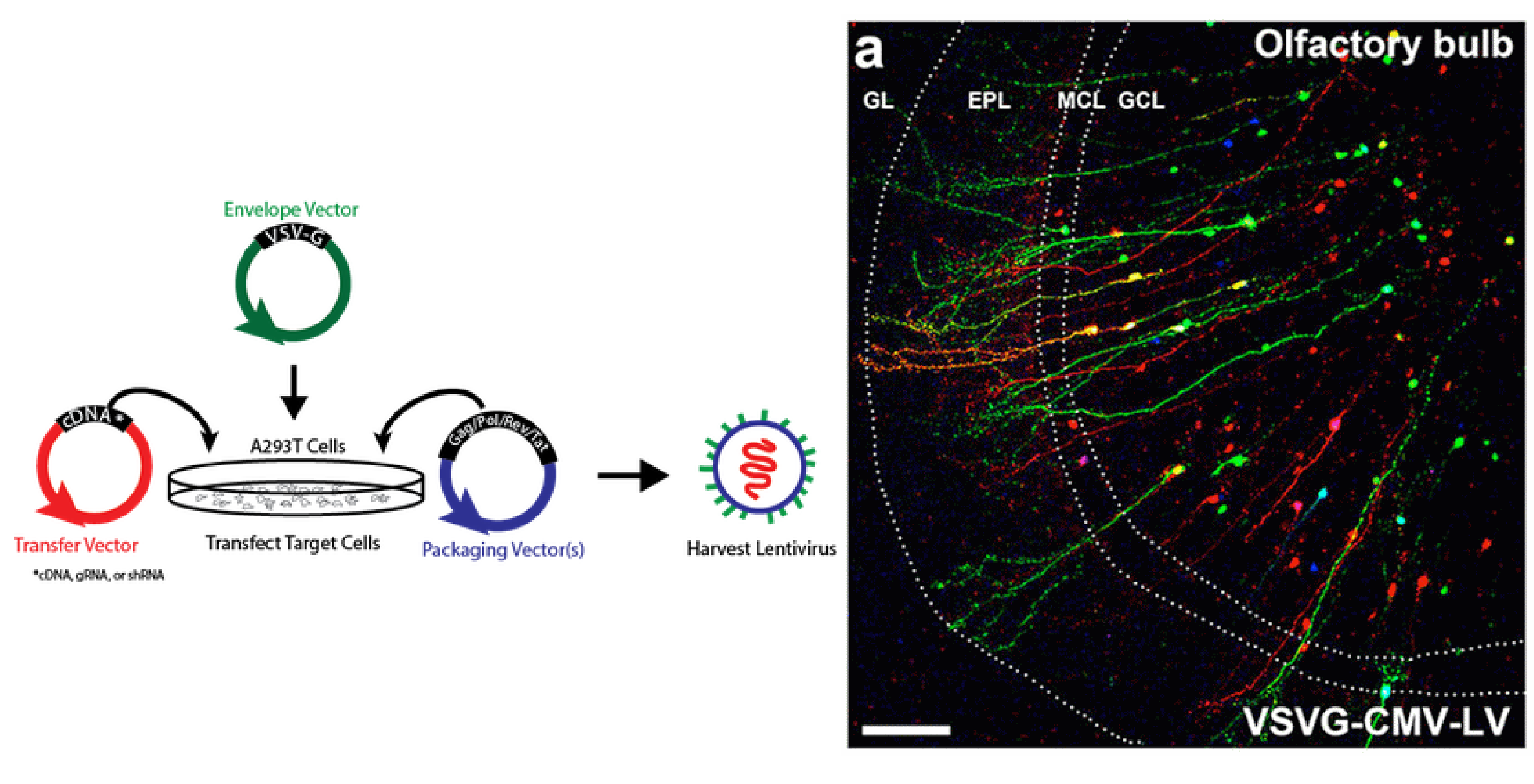
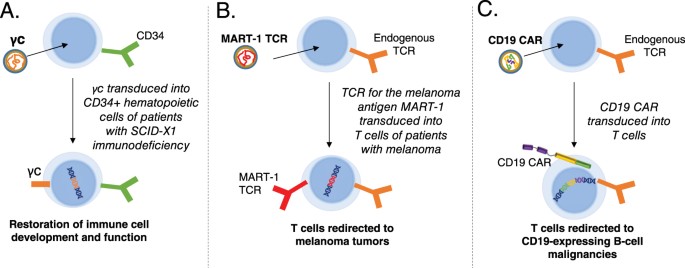
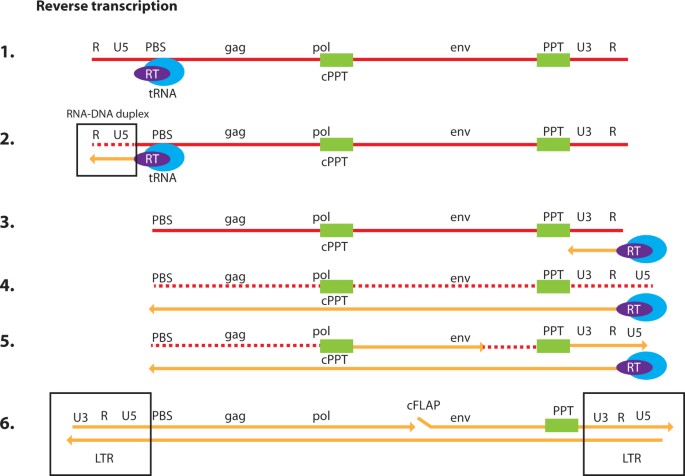
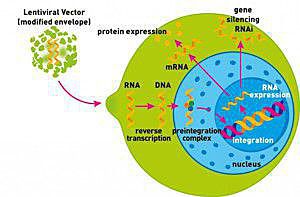
Biosafety Studies of a Clinically Applicable Lentiviral Vector for the Gene Therapy of Artemis-SCID: Molecular Therapy - Methods & Clinical Development

Concise review on optimized methods in production and transduction of lentiviral vectors in order to facilitate immunotherapy and gene therapy - ScienceDirect

Pre-clinical Safety and Efficacy of Lentiviral Vector-Mediated Ex Vivo Stem Cell Gene Therapy for the Treatment of Mucopolysaccharidosis IIIA: Molecular Therapy - Methods & Clinical Development

Rapid In-Process Monitoring of Lentiviral Vector Particles by High-Performance Liquid Chromatography: Molecular Therapy - Methods & Clinical Development

Re-structuring lentiviral vectors to express genomic RNA via cap-dependent translation: Molecular Therapy - Methods & Clinical Development

Viruses | Free Full-Text | Toward Tightly Tuned Gene Expression Following Lentiviral Vector Transduction

Promoter considerations in the design of lentiviral vectors for use in treating lysosomal storage diseases: Molecular Therapy - Methods & Clinical Development

A Guide to Approaching Regulatory Considerations for Lentiviral-Mediated Gene Therapies | Human Gene Therapy Methods
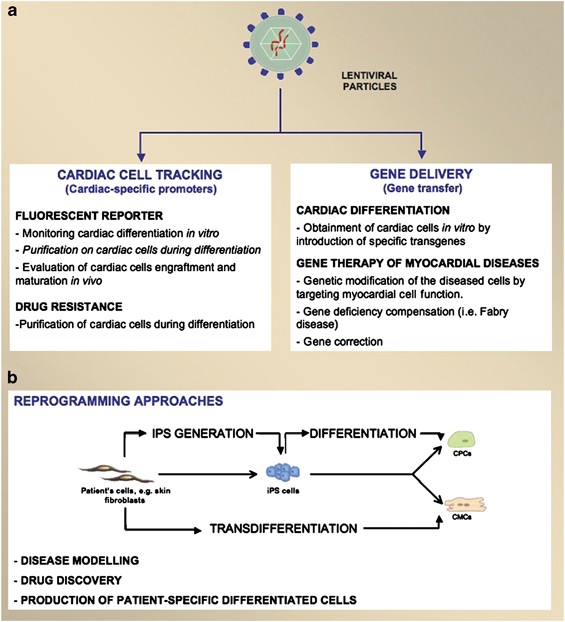
Lentiviral vectors and cardiovascular diseases: a genetic tool for manipulating cardiomyocyte differentiation and function | Gene Therapy