Incorporating Site-less Clinical Trials Into Drug Development: A Framework for Action - Clinical Therapeutics
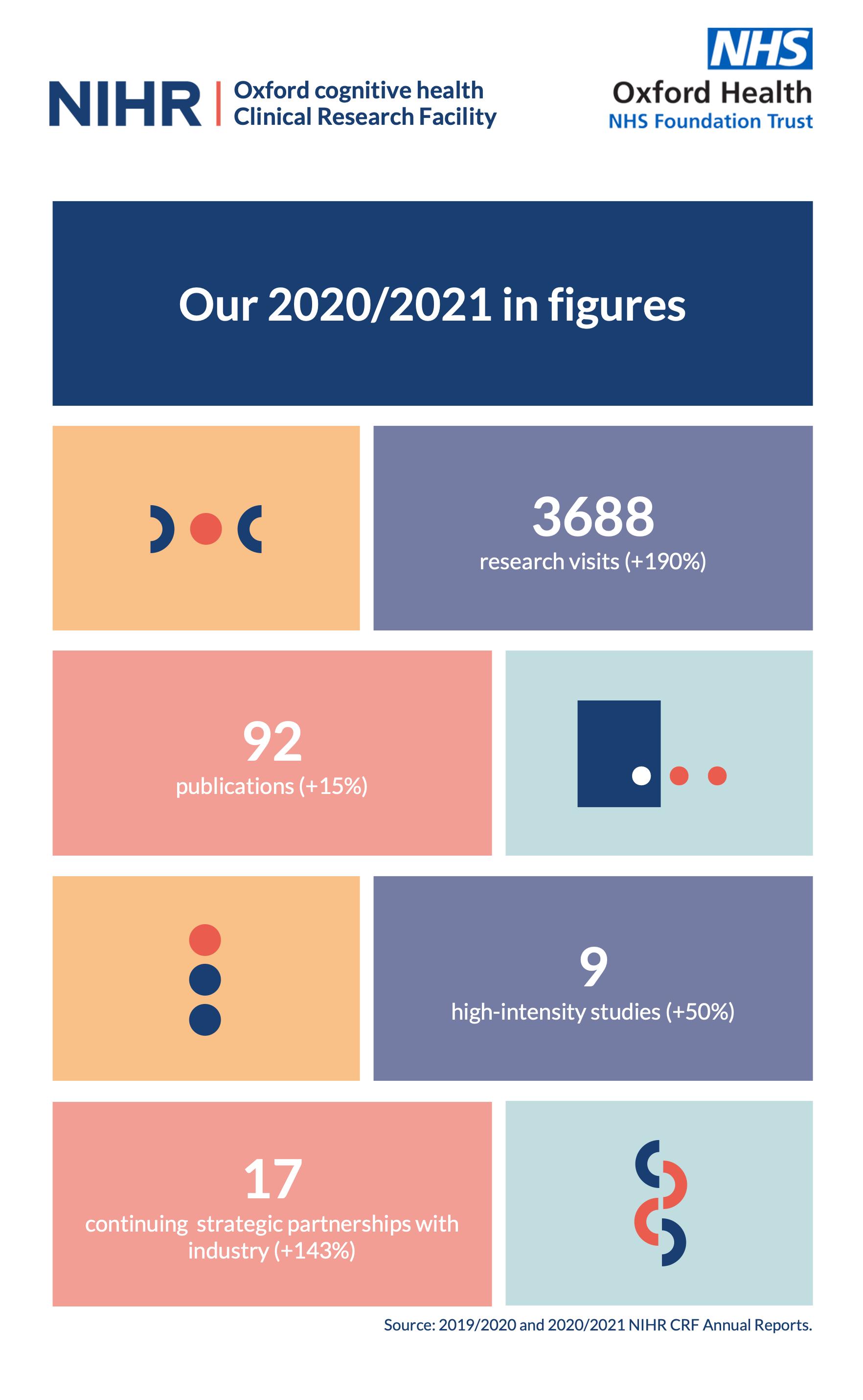
NIHR Oxford cognitive health Clinical Research Facility – NIHR Oxford Health Biomedical Research Centre
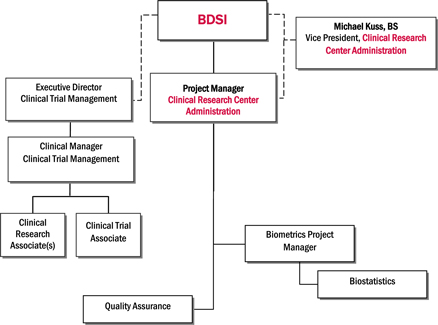
A designed clinical trial process for the Clinical Trial Management... | Download Scientific Diagram

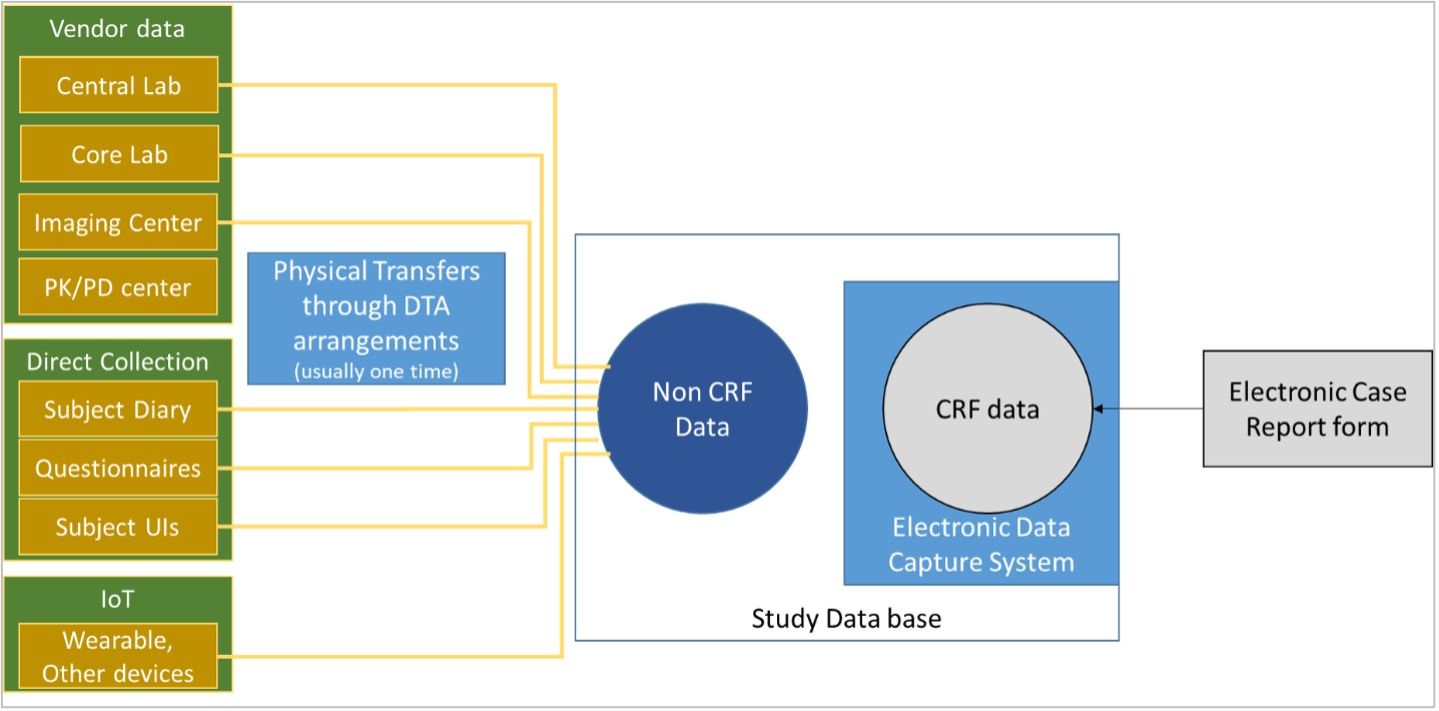
Data Management in Clinical Trials. E-CRF design / P-CRF Data entryData validation Data import Clinical coding DATABASE CLOSURE Database Closure Documents. - ppt download