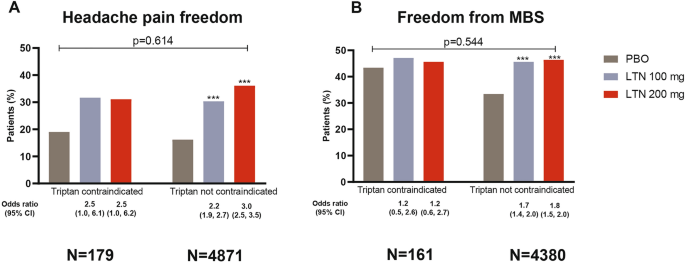
Lasmiditan for Patients with Migraine and Contraindications to Triptans: A Post Hoc Analysis | SpringerLink

Frequency of adverse events in the clinical phase III trials of lasmiditan. | Download Scientific Diagram

PDF) Sustained responses to lasmiditan: Results from post-hoc analyses of two Phase 3 randomized clinical trials for acute treatment of migraine
H8H-CD-LAHK A Study of Three Doses of Lasmiditan (50 mg, 100 mg and 200 mg) Compared to Placebo in the Acute TReaTment of MigrAi
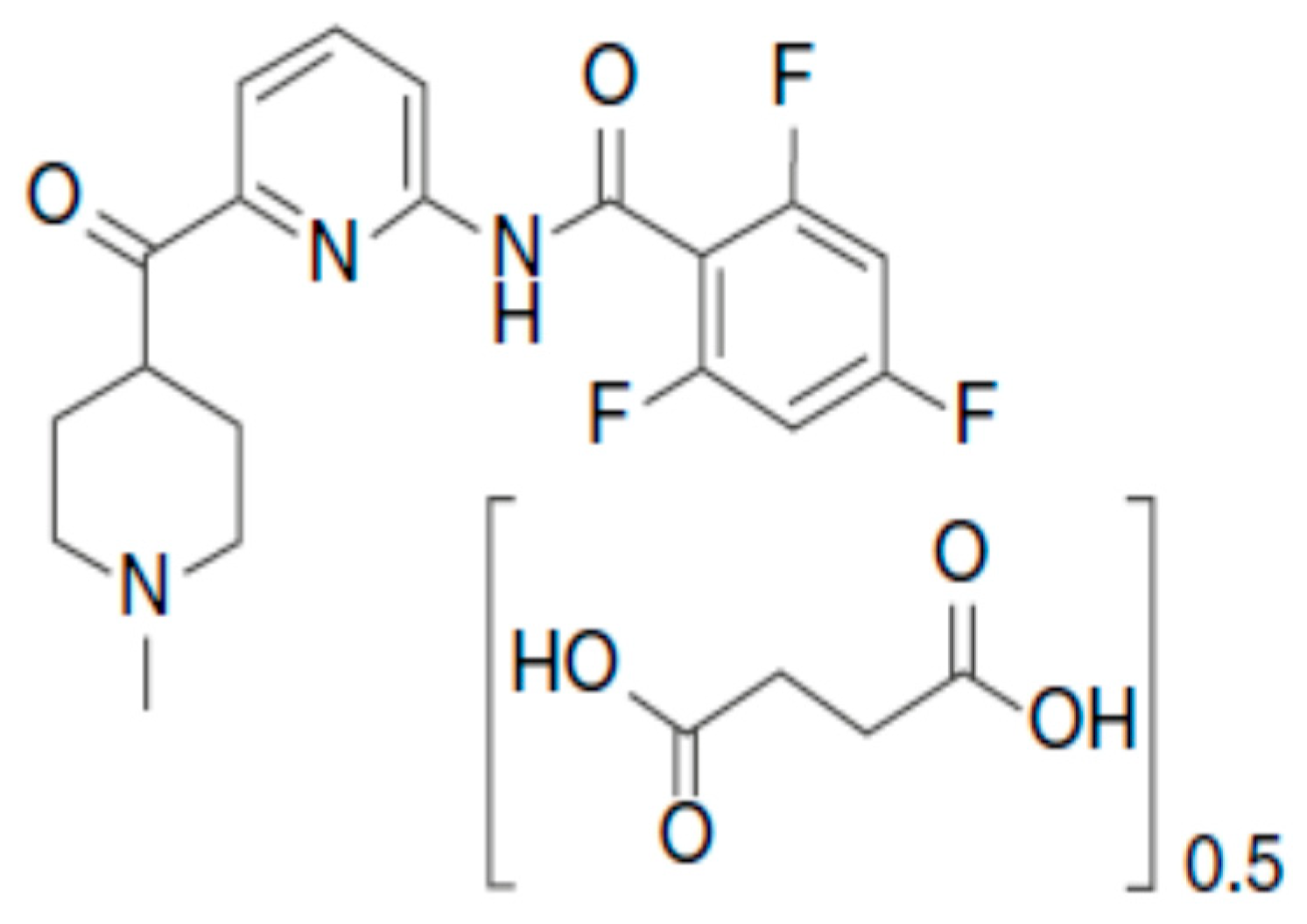
Pharmaceutics | Free Full-Text | Pharmacokinetics, Pharmacodynamics and Drug –Drug Interactions of New Anti-Migraine Drugs—Lasmiditan, Gepants, and Calcitonin-Gene-Related Peptide (CGRP) Receptor Monoclonal Antibodies | HTML
Protocol H8H-CD-LAHN (V1) (CUD-P4-001 (COL MIG-113)) A Phase I, Multicenter, Open-Label, Parallel-Group Adaptive Pharmacokinetic
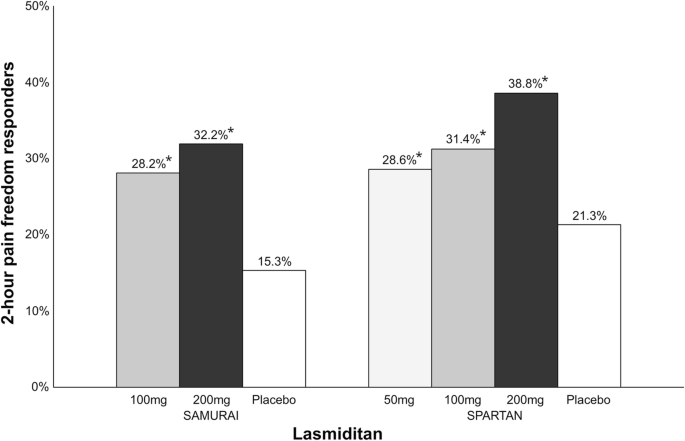
Therapeutic novelties in migraine: new drugs, new hope? | The Journal of Headache and Pain | Full Text
The pharmacological profile and clinical prospects of the oral 5-HT1F receptor agonist lasmiditan in the acute treatment of migr

Lasmiditan Col144 Investigational Drug Treatment Acute Stock Vector (Royalty Free) 1332679202 | Shutterstock
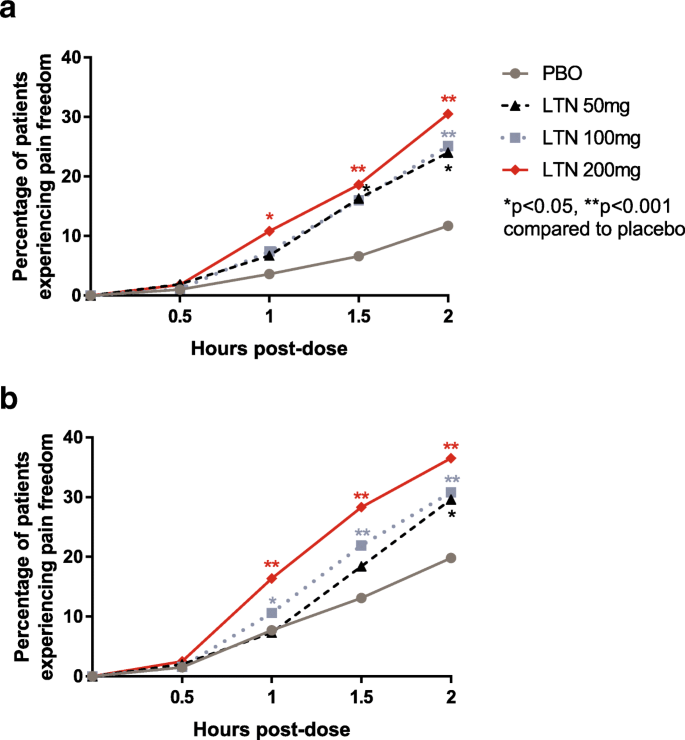
Efficacy and safety of lasmiditan in patients using concomitant migraine preventive medications: findings from SAMURAI and SPARTAN, two randomized phase 3 trials | The Journal of Headache and Pain | Full Text









