The statistical content of published medical research: some implications for biomedical education – Pubrica Academy

Amazon - Statistical Design and Analysis of Clinical Trials: Principles and Methods (Chapman & Hall/CRC Biostatistics Series): Shih, Weichung Joe, Aisner, Joseph: 9781482250497: Books

The Statistician's view of a Clinical Trial | 40 years of Clinical Research Statistical Analysis & Clinical Trial Design expertise

Methods and Applications of Statistics in Clinical Trials, Volume 1: Concepts, Principles, Trials, and Designs | Wiley

Role of Biostatistics and Responsibilities of Biostatisticians in Clinical Medical Research – Academy

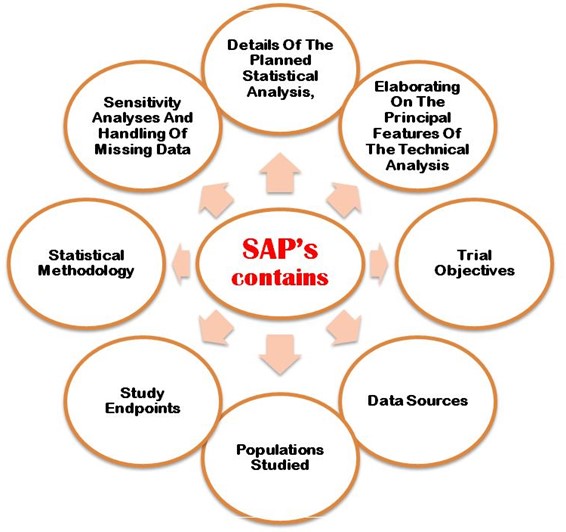
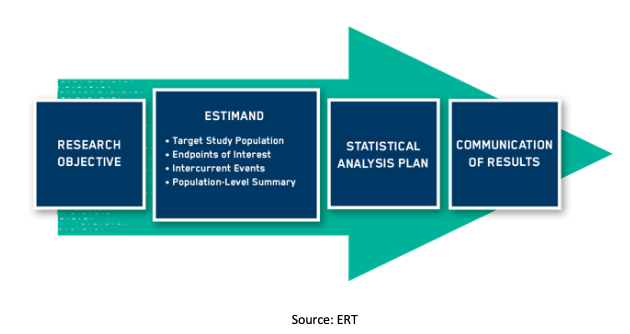
Early phase clinical trials extension to guidelines for the content of statistical analysis plans | The BMJ

Impact of trial design on the estimation of drug potency and power in clinical trials of haemophilia with inhibitors - ScienceDirect

Introduction to Statistical Methods for Clinical Trials (Chapman & Hall/CRC Texts in Statistical Science) 1, Cook, Thomas D., DeMets, David L. - Amazon.com

Detailed statistical analysis plan for the pulmonary protection trial – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on CyberLeninka open science hub.

Statistical methods to evaluate the effectiveness of cancer treatments in clinical trials | STAT | Statistics

A Technology-Driven Solution for Optimized Statistical Analysis of Clinical Trial Biomarker Data - Quartz Bio

PDF) Statistical methods for use in the analysis of randomized clinical trials utilizing a pretreatment, posttreatment, follow-up (PPF) paradigm
![PDF] A framework for the pre-specification of statistical analysis strategies in clinical trials (Pre-SPEC) | Semantic Scholar PDF] A framework for the pre-specification of statistical analysis strategies in clinical trials (Pre-SPEC) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d44e12934bbb4b1d0a68121536610fdef42bd628/9-Table1-1.png)
PDF] A framework for the pre-specification of statistical analysis strategies in clinical trials (Pre-SPEC) | Semantic Scholar